15/07/2022
ISO 13485 is an international standard issued by the International Organization for Standardization (ISO) concerning the quality management system for medical products. It aims to ensure that products are provided in compliance with regulatory requirements and customer needs.
ISO 13485:2016 applies to organizations that manufacture and supply medical devices and related services in the healthcare sector. Medical products that may obtain certification include face masks, gloves, needles, infusion sets, sterilization solutions, feeding tubes, etc.

The healthcare sector directly impacts human health and therefore requires strict management. When an organization applies ISO 13485, it can bring many benefits such as:
Improving product quality and ensuring user safety.
Creating competitive advantages and enhancing brand professionalism, even supporting export opportunities.
Increasing revenue and profit, saving costs, and minimizing risks.
Establishing a management system that facilitates quality control and improves production efficiency.
Meeting mandatory requirements of the industry and legal regulations.
Facilitating integration with other management systems such as ISO 14001, ISO 9001, and ISO/IEC 17025.

ISO 13485:2003 was first issued by the international organization in July 2003. ISO 13485:2016 is the latest version replacing the 2003 edition. Both standards are equivalent in value; however, the 2016 version includes additional provisions related to risk management principles.
The decision to apply ISO 13485:2016 and ISO 9001:2015 depends entirely on the enterprise and the products it provides. If an enterprise both supplies medical devices and markets research products or medical equipment, dual certification is recommended.
This is because ISO 9001 certification also relates to industrial or general products. Moreover, when applying both certifications simultaneously, the review of legal documentation and procedures can be conducted at the same time.

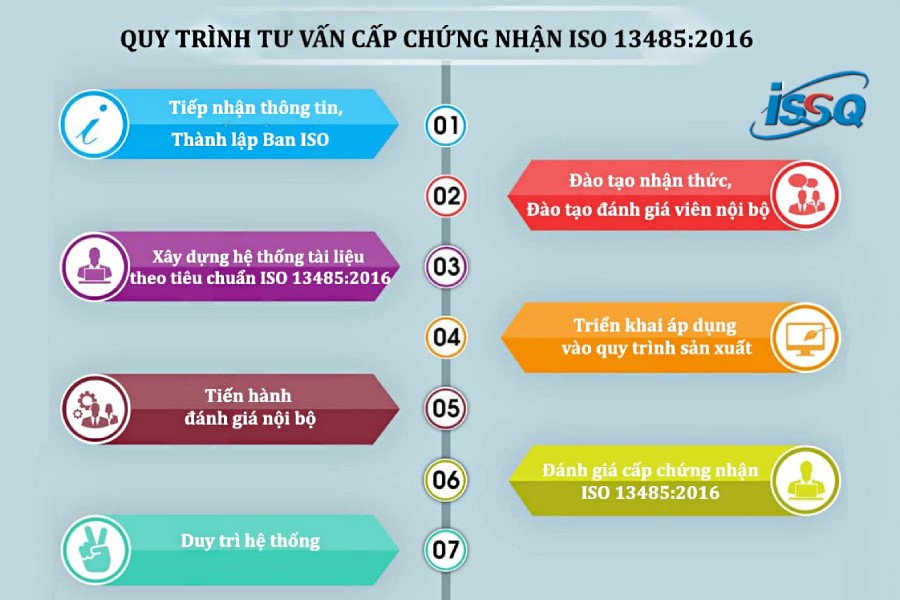
To ensure that an enterprise obtains ISO 13485:2016 certification, the implementation process typically follows these steps:
Receiving information and establishing an ISO 13485 project team.
Conducting awareness training and internal auditor training.
Developing a documentation system in accordance with ISO 13485 requirements.
Implementing the system in production processes.
Carrying out internal audits.
Certification audit for ISO 13485.
Maintaining and improving the system.
Currently, many organizations provide ISO 13485 certification services; however, not all are reputable. Enterprises should therefore select organizations that:
Are designated and competent to conduct ISO 13485 certification assessments.
Are licensed to operate by competent state authorities.
Possess consultants and experts with relevant qualifications.
If your enterprise is seeking a reputable ISO certification body, you may refer to ISSQ Institute of Quality (ISSQ Quality Research Institute). With a team of experienced experts, ISSQ was established under Decision No. 233/QĐ-LHHVN of the Vietnam Union of Science and Technology Associations.
For ISO 13485:2016 certification needs, please contact ISSQ Institute of Quality.
ISSQ is always ready to accompany enterprises during integration and development.
Posting date: 15/07/2022